BREAKING! Scientists Discover Ancient Protein Motif That Sculpts RNA and Triggers Diseases from Cancer to Brain Disorders!
Nikhil Prasad Fact checked by:Thailand Medical News Team Apr 04, 2025 3 weeks, 6 hours, 22 minutes ago
Medical News: Scientists Decode a Molecular Fossil That Shapes Life Itself
In a groundbreaking discovery that bridges molecular biology, evolution, and human disease, researchers from The University of Texas MD Anderson Cancer Center and Lawrence Berkeley National Laboratory have uncovered the secrets of a tiny but powerful structural element hidden within thousands of life-sustaining proteins. This finding could revolutionize our understanding of how genes are expressed, how cells communicate, and even how deadly diseases take hold in the human body.
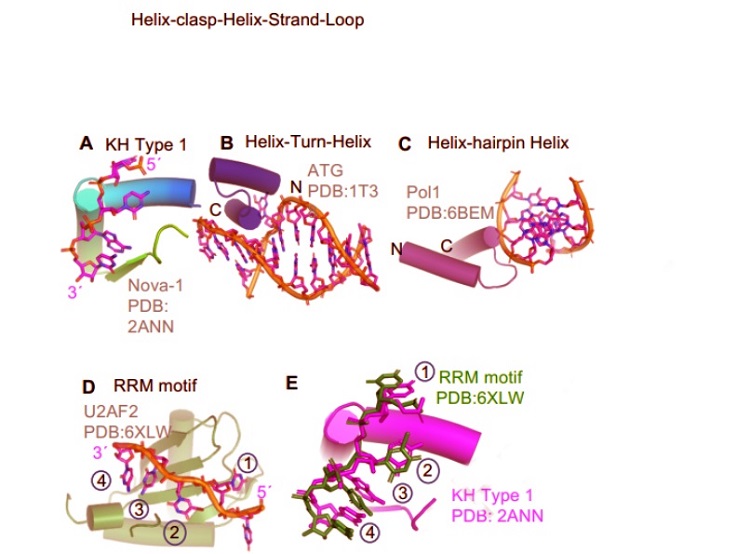 The HcH–SL motif is distinct from other known helical and strand nucleic acid recognition motifs. A. In Type I HcH–SL, the protein helices form a right angle, righthanded corner and together with the strand-loop, sculpts the nucleic acid and enforces ssNA specificity. B. Helix-Turn-Helix motifs recognize the duplex DNA helical nature and insert one protein helix into the major or minor groove, distorting the DNA. The protein helices cross each other and are roughly orthogonal. C. Helix-hairpin-Helix binds to one strand of helical duplex DNA. The protein helices cross each other at ~30° angle. D. In RNA Recognition motif (RRM), the beta strands are the major RNA-interaction interface. E. The first four nts bound by the RRM are splayed in similar orientation as the four nts bound by the KH domain. The HcH–SL of the KH domain is shown for reference.
The HcH–SL motif is distinct from other known helical and strand nucleic acid recognition motifs. A. In Type I HcH–SL, the protein helices form a right angle, righthanded corner and together with the strand-loop, sculpts the nucleic acid and enforces ssNA specificity. B. Helix-Turn-Helix motifs recognize the duplex DNA helical nature and insert one protein helix into the major or minor groove, distorting the DNA. The protein helices cross each other and are roughly orthogonal. C. Helix-hairpin-Helix binds to one strand of helical duplex DNA. The protein helices cross each other at ~30° angle. D. In RNA Recognition motif (RRM), the beta strands are the major RNA-interaction interface. E. The first four nts bound by the RRM are splayed in similar orientation as the four nts bound by the KH domain. The HcH–SL of the KH domain is shown for reference.
This
Medical News report delves into the remarkable Helix-clasp-Helix–Strand-Loop motif, or HcH–SL - a structure so ancient and versatile that scientists believe it may have existed in the earliest forms of life, long before DNA became the dominant genetic material. Led by Dr. John A. Tainer and Dr. Susan E. Tsutakawa, the team revealed how this unique motif enables a critical family of proteins, known as KH domains, to sculpt RNA molecules with extraordinary precision. Their findings connect a molecular structure to everything from cancer and cardiovascular disease to neurological and developmental disorders.
What Are KH Domains and Why Do They Matter
The KH (K Homology) domain is a protein segment first identified in a human protein that binds RNA. Since its discovery, it has been found in more than 233,000 proteins across bacteria, archaea, and humans. These domains are essential for controlling RNA, the molecular cousin of DNA responsible for carrying genetic instructions and orchestrating key processes like gene expression, protein synthesis, and cell signaling.
KH domains work by binding to single-stranded RNA or DNA, recognizing specific sequences and chemical structures. What makes this discovery exciting is the new structural insight: a tiny architectural feature within KH domains, the HcH–SL motif, gives them the power to identify and manipulate RNA at an atomic level.
The Sculpting Power of the HcH–SL Motif
The HcH–SL motif is a structural triangle made from two helices, a beta strand, and a loop, all held together by a special sequence of amino acids inc
luding glycine residues. It forms a right-angled, right-handed triangle that acts like a microscopic clamp or "hand" that not only binds RNA but reshapes it.
RNA molecules are made of chains of nucleotides - think of beads on a string - but when they bind to a KH domain equipped with this motif, their bases become unstacked and splayed out, like cards fanned in a magician’s hand. This unique positioning lets the protein read the RNA's chemical features in exquisite detail, recognizing both its sequence and any chemical modifications - like methyl groups - that may be present.
What’s more, this structural motif doesn’t just bind RNA - it sculpts it. By altering how RNA bases are presented, the HcH–SL motif enables other proteins to interact with it more efficiently or accurately. It is a kind of molecular pre-processing step critical for fine-tuning gene expression and RNA-related tasks.
Link to Diseases and Viral Hijacking
The implications of this RNA sculpting go far beyond molecular biology. Errors in KH domain proteins—often due to mutations or altered expression - are now known to be linked with a long list of human diseases.
For instance, Fragile X syndrome, a leading genetic cause of intellectual disability, has been traced to a single mutation in the KH domain of the FMR1 protein.
Similarly, abnormal KH domain activity has been observed in many cancers, including breast, liver, kidney, colorectal, and prostate cancer, as well as in neurodevelopmental and cardiovascular disorders.
In many of these cases, it is the disruption of RNA recognition or RNA sculpting that causes critical biological pathways to fail. Some KH proteins are known to control tumor suppressor microRNAs, regulate DNA repair, or stabilize cancer-related RNAs.
Even more remarkably, some viruses - including SARS-CoV-2 - appear to have evolved peptides that mimic RNA and bind directly to KH domains, hijacking them for viral replication. This raises the possibility that KH domains could be drug targets to prevent such viral exploitation.
An Evolutionary Relic from the Origins of Life
What makes the HcH–SL motif even more intriguing is its potential role in the early evolution of life. Researchers suggest it may have existed in the Last Universal Common Ancestor (LUCA), the ancient cell type that gave rise to all known life on Earth. This motif likely played a key role in early RNA replication and regulation, before the emergence of DNA-based life.
Unlike many protein domains that vary across species, the structure and function of HcH–SL are preserved in both bacterial and human KH domains. This suggests the motif’s design is not only ancient but nearly perfect for its role. It’s also one of the rare structures able to detect changes in the chemistry of RNA bases, including modifications that can signal environmental changes, stress, or even disease.
How It Works at the Molecular Level
By studying over 40 high-resolution protein - RNA complexes, the scientists showed that the HcH–SL motif interacts with RNA through multiple contact points. One helix unpacks the bases, the clasp stabilizes the triangle, the second helix provides an electrostatic anchor, and the beta strand plus its loop recognize the trailing RNA bases. These interactions are not random - they are highly organized and conserved.
The motif can bind to both RNA and DNA, but certain KH domains are RNA-specific, often depending on the presence of RNA-specific chemical groups like the 2’ hydroxyl. In cancer biology, for example, some KH domains preferentially bind to methylated RNA - a chemical modification known to play roles in cancer progression and therapy resistance.
One example is the family of proteins called IGF2BPs, which use their KH domains to recognize methylated RNA and stabilize cancer-promoting transcripts. They even change their RNA-binding conformation in response to methylation - a feat enabled by the flexibility of the HcH–SL motif.
Why This Matters for Future Medicine and Biotechnology
The discovery of the HcH–SL motif’s sculpting function opens new doors in medicine and synthetic biology. By targeting this motif with drugs or designing artificial KH proteins, scientists may one day be able to correct genetic miscommunications in diseases like Fragile X syndrome or cancers with KH domain dysregulation.
There is also strong potential for using engineered HcH–SL motifs to create synthetic RNA readers for diagnostics or for regulating gene expression in RNA-based therapies and vaccines. In agriculture, KH proteins could be tweaked to improve stress responses in plants or make them resistant to viral infections.
The motif’s structural elegance and specificity make it an ideal component for synthetic molecular machines or sensors - possibly even chimeric proteins that could detect viral RNA in real time or regulate vaccine responses.
Conclusion
The identification and structural decoding of the HcH–SL motif mark a major milestone in our understanding of RNA biology and protein evolution. It not only reveals how ancient proteins recognize and reshape RNA with surgical precision, but it also illuminates how malfunctions in this process can trigger a wide range of human diseases. Perhaps most excitingly, the HcH–SL motif stands as a relic from the dawn of life—an evolutionary survivor whose subtle geometry still influences the machinery of modern cells. As science uncovers more about how it works, this unassuming triangle may provide powerful new tools to combat disease, enhance biotechnology, and illuminate the very origins of life itself.
The study findings were published in the peer reviewed Journal of Biological Chemistry.
https://www.sciencedirect.com/science/article/pii/S0021925825003230
For the latest on Proteomics, keep on logging to Thailand
Medical News.
Read Also:
https://www.thailandmedical.news/news/breaking-thousands-of-novel-hybrid-genes-or-chimeric-rnas-found-in-the-blood-of-the-covid-19-infected
https://www.thailandmedical.news/news/french-scientists-discover-that-sars-cov-2-envelope-protein-s-pdz-binding-motif-disrupts-host-s-epithelial-cell-cell-junction
https://www.thailandmedical.news/news/covid-19-news-french-study-finds-that-sars-cov-2-envelop-protein-has-a-carboxy-terminal-that-contains-a-pdz-binding-motif-that-is-pathogenetic
https://www.thailandmedical.news/articles/hospital-news
https://www.thailandmedical.news/pages/thailand_doctors_listings
