BREAKING! University of California Study Alarmingly Confirms That SARS-CoV-2 Coronavirus Can Infect And Replicate In Human Host Neurons!
Source: NEUROCOVID Nov 23, 2021 4 years, 4 months, 1 week, 4 days, 19 hours, 26 minutes ago
NEUROCOVID: For a while despite past studies indicating that the SARS-CoV-2 coronavirus could infect the neurons and that was the cause of numerous neurological and also psychiatric issues arising in COVID-19 patients and also in long COVID, there was still much debate among the scientific community and the need for further concrete evidence.
A new study by researchers from the University of California-Irvine along with scientists from the Scripps Research Institute-California has confirmed that the SARS-CoV-2 coronavirus is able to infect and replicate in the human host neurons.
Infections with SARS-CoV-2, the viral pathogen responsible for COVID-19, are often associated with neurologic symptoms that range from mild to severe, yet increasing evidence argues the virus does not exhibit extensive neuroinvasive properties.
The study team demonstrated that the SARS-CoV-2virus can infect and replicate in human iPSC-derived neurons and that infection shows limited anti-viral and inflammatory responses but increased activation of EIF2 signaling following infection as determined by RNA sequencing.
In the study, intranasal infection of K18 human ACE2 transgenic mice (K18-hACE2) with SARS-CoV-2 virus resulted in lung pathology associated with viral replication and immune cell infiltration.
Furthermore, about 50% of infected mice exhibited CNS infection characterized by wide-spread viral replication in neurons accompanied by increased expression of chemokine (
Cxcl9, Cxcl10, Ccl2, Ccl5 and
Ccl19) and cytokine (
Ifn-λ and
Tnf-α) transcripts associated with microgliosis and a neuroinflammatory response consisting primarily of monocytes/macrophages.
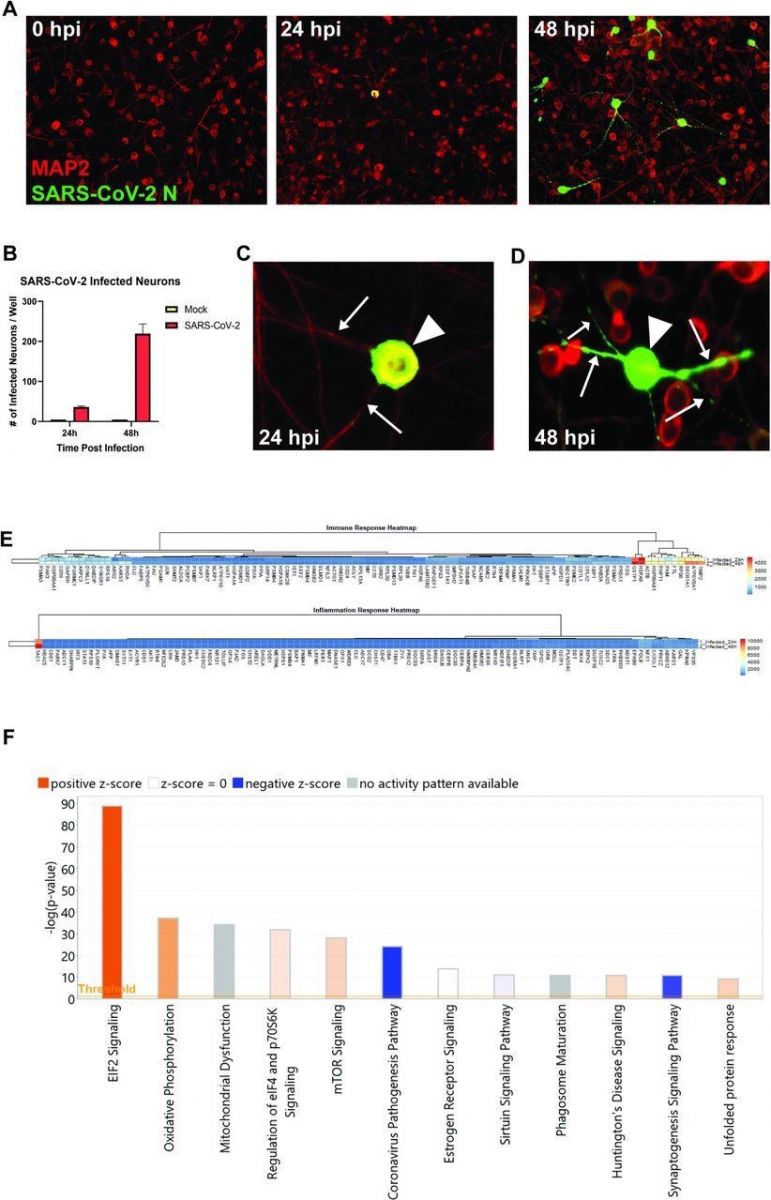 SARS-CoV-2 infects human iPSC-derived neurons. (A) hiPSC-derived neurons were infected with SARS-CoV-2 at an MOI of 0.1, immunostained with anti-MAP2 and anti-SARS-CoV-2 N, and imaged at 0, 24, and 48 hours post-infection. (B) Quantification of SARS-CoV-2 GFP fluorescence of mock-infected and SARS-CoV-2-infected hiPSC-derived neurons. (C) Perinuclear replication of SARS-CoV-2 in neuronal soma (arrowhead) but no viral axonal (arrows) transport at 24 hours post-infection. (D) Perinuclear presence of SARS-CoV-2 in soma (arrowhead) and axon (arrows) at 24 hours post-infection. (E) Heat map of genes expressed 24 and 48h post-infection. (F) Top 12 canonical pathways showing progressive changes from 24 to 48 h post-infection.
SARS-CoV-2 infects human iPSC-derived neurons. (A) hiPSC-derived neurons were infected with SARS-CoV-2 at an MOI of 0.1, immunostained with anti-MAP2 and anti-SARS-CoV-2 N, and imaged at 0, 24, and 48 hours post-infection. (B) Quantification of SARS-CoV-2 GFP fluorescence of mock-infected and SARS-CoV-2-infected hiPSC-derived neurons. (C) Perinuclear replication of SARS-CoV-2 in neuronal soma (arrowhead) but no viral axonal (arrows) transport at 24 hours post-infection. (D) Perinuclear presence of SARS-CoV-2 in soma (arrowhead) and axon (arrows) at 24 hours post-infection. (E) Heat map of genes expressed 24 and 48h post-infection. (F) Top 12 canonical pathways showing progressive changes from 24 to 48 h post-infection.
Microglia depletion via administration of colony-stimulating factor 1 receptor inhibitor, PLX5622, in SARS-CoV-2 infected mice did not affect survival or viral replication but did result in dampened expression of proinflammatory cytokine/chemokine transcripts and a reduction in monocyte/macrophage infiltration.
The study findings argue that microglia are dispensable in terms of controlling SARS-CoV-2 replication in in the K18-hACE2 model but do contribute to an inflammatory response through expression of pro-inflammatory genes.
The
tps://www.thailandmedical.news/news/neurocovid-study-shows-that-even-moderate-covid-19-cases-experience-long-term-neurological-complications-and-harm">NEUROCOVID study findings contribute to previous work demonstrating the ability of SARS-CoV-2 to infect human host neurons as well as emphasizing the potential use of the K18-hACE2 model to study immunological and neuropathological aspects related to SARS-CoV-2-induced neurologic disease.
The study findings were published on a preprint server and are currently being peer reviewed.
https://www.biorxiv.org/content/10.1101/2021.11.15.468761v1
Conditions of encephalitis and meningitis have been reported in several COVID-19 patients, and viral RNA and protein have also been identified within the CSF of infected patients. Although human brain organoids are vulnerable to severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection, extensive CNS penetrance by SARS-CoV-2 has not been demonstrated.
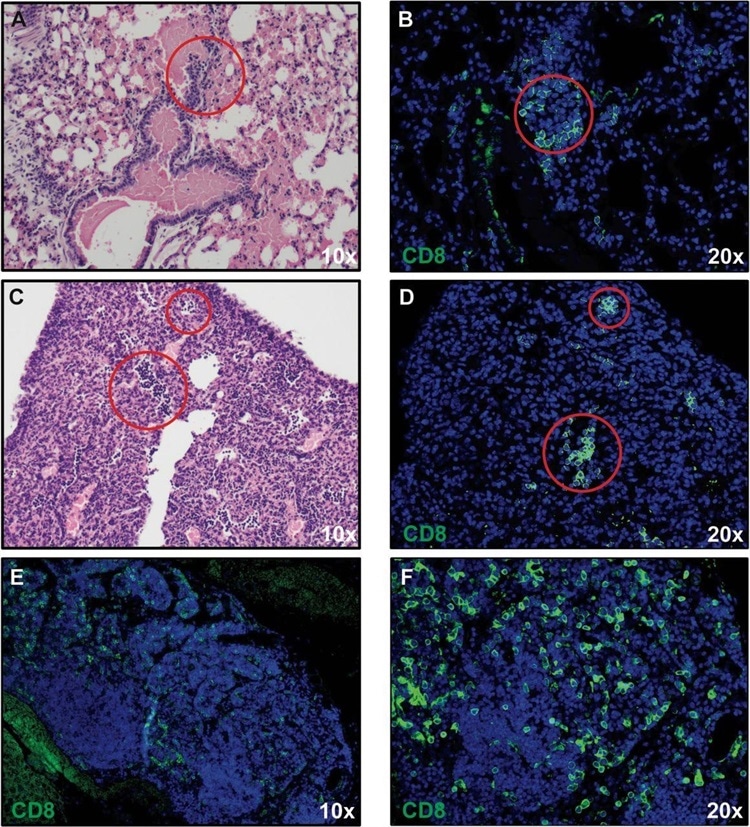 CD8+ T cell infiltration into lungs of SARS-CoV-2-infected mice. H&E staining of lungs of SARS-CoV-2 infected mice at day 7 post-infection reveal inflammation (A and C) associated with CD8+ T cell infiltration (B and D) as determined by immunofluorescent staining. Lymph node-like structures were also detected containing CD8+ T cells (E and F). Panels A, C, and E 10X magnification; panels B, D, and F 20X magnification.
CD8+ T cell infiltration into lungs of SARS-CoV-2-infected mice. H&E staining of lungs of SARS-CoV-2 infected mice at day 7 post-infection reveal inflammation (A and C) associated with CD8+ T cell infiltration (B and D) as determined by immunofluorescent staining. Lymph node-like structures were also detected containing CD8+ T cells (E and F). Panels A, C, and E 10X magnification; panels B, D, and F 20X magnification.
However pre-clinical animal models such as human angiotensin-converting enzyme 2 (hACE2) transgenic mouse models can provide key insights into COVID-19 pathogenesis.
The study team utilized the intranasal SARS-CoV-2 infection of K18-hACE2 transgenic mice to study the neurological impact of SARS-CoV-2.
The study team attempted to further increase the scope of previous studies in examining SARS-CoV-2 infection of human CNS resident cells.
The team evaluated the immune response to SARS-CoV-2 infection of the CNS of K18-hACE2 mice and assessed the influence of microglia in host defense following CNS infection by SARS-CoV-2.
For the study, the human iPSC-derived neurons were inoculated with SARS-CoV-2.
Alarmingly, the staining of nucleocapsid proteins confirmed that SARS-CoV-2 was able to infect and replicate within neurons.
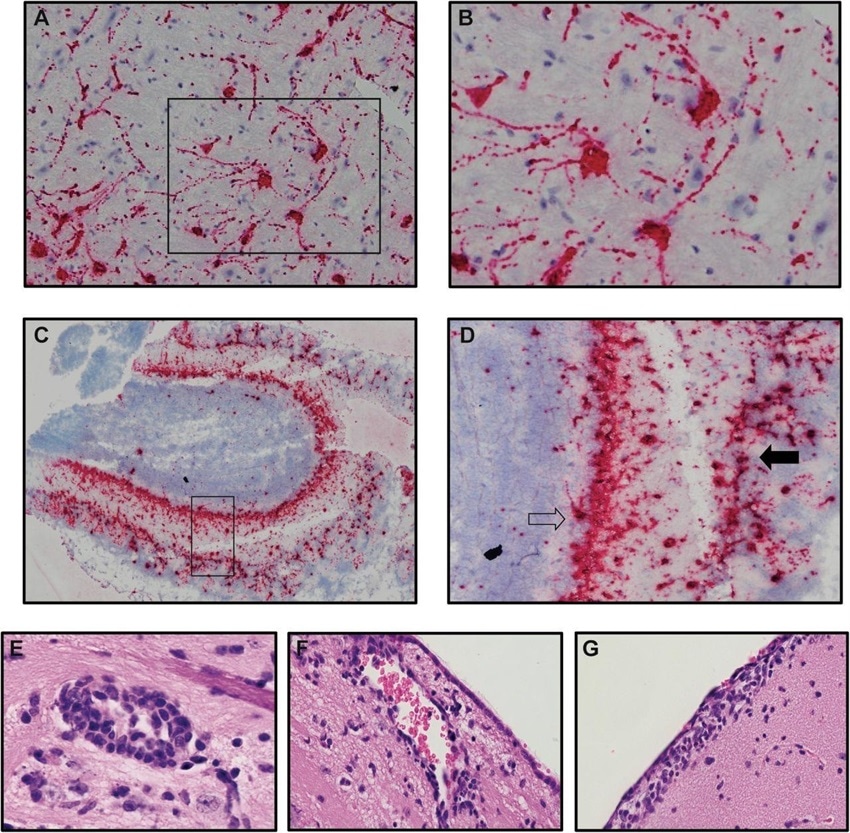 Neurons are targets of infection within the brains of SARS-CoV-2 infected K18-hACE2 mice. Brains of SARS-CoV-2 infected mice at day 7 p.i. were analyzed to assess cellular targets of infection through in situ hybridization using RNAscope in situ hybridization using Spike-specific probes. (A) Cells within the cortex with neuron morphology were primary targets of infection; (B) high-power image of cells boxed in panel A show viral RNA present within cell body as well as extending down dendrites extending from the cell body. (C) Viral RNA was also detected in olfactory bulbs at day 7 p.i. (D) high-power image cells boxed in panel C reveal neurons in the mitral (open arrow) and glomerular (closed arrow) are infected by virus. Representative H&E images from the brains of infected K18-hACE2 mice at day 7 p.i. depicting (E) perivascular cuffing, (F) subventricular inflammation, and (G) leptomeningitis.
Neurons are targets of infection within the brains of SARS-CoV-2 infected K18-hACE2 mice. Brains of SARS-CoV-2 infected mice at day 7 p.i. were analyzed to assess cellular targets of infection through in situ hybridization using RNAscope in situ hybridization using Spike-specific probes. (A) Cells within the cortex with neuron morphology were primary targets of infection; (B) high-power image of cells boxed in panel A show viral RNA present within cell body as well as extending down dendrites extending from the cell body. (C) Viral RNA was also detected in olfactory bulbs at day 7 p.i. (D) high-power image cells boxed in panel C reveal neurons in the mitral (open arrow) and glomerular (closed arrow) are infected by virus. Representative H&E images from the brains of infected K18-hACE2 mice at day 7 p.i. depicting (E) perivascular cuffing, (F) subventricular inflammation, and (G) leptomeningitis.
Interestingly the study observations suggested that the SARS-CoV-2 virus may not spread via fusion with neighboring cells since syncytia formation was not detected in neuron cultures. Although the Coronavirus Pathogenesis Pathway was overrepresented, it was inhibited in response to neuronal infection by SARS-CoV-2.
For the study, K18-hACE2 mice were intranasally infected with plaque-forming units (PFU) of SARS-CoV-2, and the resulting weight loss and mortality were recorded.
The study findings showed the development of interstitial pneumonia and immune cell infiltration associated with viral RNA present within the lungs.
Furthermore, the expression of proinflammatory cytokines (Ifn-λ and Tnf-α)/chemokines (Cxcl9, Cxcl10, Ccl2, Ccl5, and Ccl19) in response to infection increased, which correlated with microgliosis and the presence of inflammatory cells.
The study team identified inflammatory CD8+ T cells in the lungs of infected mice, which were presumably responding to the T cell chemoattractant CXCL10.
It was also found that inflammatory monocyte/macrophages were likely employed in response to the expression of CCL2. The presence of inflammatory neutrophils was likely reflected due to the increased expression of transcripts encoding CXCR2.
Interestingly, the study found that, in SARS-CoV-2-infected mice, microglia depletion via administration of colony-stimulating factor 1 receptor inhibitor PLX5622 did not affect survival or viral replication.
The expression of proinflammatory cytokine/chemokine transcripts was however dampened, and a reduction in monocyte/macrophage infiltration was found.
Corresponding author, Dr Thomas E. Lane, Ph.D. from the Department of Neurobiology & Behavior, School of Biological Sciences at the University of California-Irvine told Thailand
Medical News, “Importantly the study findings show that microglia did not have a role in SARS-CoV-2 replication in the K18-hACE2 model but contributed to an inflammatory response through the expression of proinflammatory genes.”
Typically, microglia play an important role in host defense in response to viral infection of the CNS.
The ablation of microglia through CSF1R inhibition causes increased mortality in mice infected with West Nile Virus (WNV) and was correlated with weakened activation of antigen-presenting cells (APCs) and limited reactivation of virus-specific T cells that leads to reduced viral clearance.
Interestingly however this study showed that microglia depletion in SARS-CoV-2 infected mice did not have an impact on viral survival or replication but did lead to dampened expression of proinflammatory cytokine/chemokines and reduced monocyte/macrophage infiltration.
Importantly the research data confirms the ability of SARS-CoV-2 to infect human host neurons and highlight the potential use of the K18-hACE2 model to study immunological and neuropathological aspects related to SARS-CoV-2-induced neurologic disease.
Dr Lane further added, “These study findings emphasize the importance of working with animal models in which SARS-CoV-2 entry into the CNS is more consistent with what has been observed in COVID-19 patients.”
For the latest on
NEUROCOVID, keep on logging to Thailand Medical News.

 SARS-CoV-2 infects human iPSC-derived neurons. (A) hiPSC-derived neurons were infected with SARS-CoV-2 at an MOI of 0.1, immunostained with anti-MAP2 and anti-SARS-CoV-2 N, and imaged at 0, 24, and 48 hours post-infection. (B) Quantification of SARS-CoV-2 GFP fluorescence of mock-infected and SARS-CoV-2-infected hiPSC-derived neurons. (C) Perinuclear replication of SARS-CoV-2 in neuronal soma (arrowhead) but no viral axonal (arrows) transport at 24 hours post-infection. (D) Perinuclear presence of SARS-CoV-2 in soma (arrowhead) and axon (arrows) at 24 hours post-infection. (E) Heat map of genes expressed 24 and 48h post-infection. (F) Top 12 canonical pathways showing progressive changes from 24 to 48 h post-infection.
SARS-CoV-2 infects human iPSC-derived neurons. (A) hiPSC-derived neurons were infected with SARS-CoV-2 at an MOI of 0.1, immunostained with anti-MAP2 and anti-SARS-CoV-2 N, and imaged at 0, 24, and 48 hours post-infection. (B) Quantification of SARS-CoV-2 GFP fluorescence of mock-infected and SARS-CoV-2-infected hiPSC-derived neurons. (C) Perinuclear replication of SARS-CoV-2 in neuronal soma (arrowhead) but no viral axonal (arrows) transport at 24 hours post-infection. (D) Perinuclear presence of SARS-CoV-2 in soma (arrowhead) and axon (arrows) at 24 hours post-infection. (E) Heat map of genes expressed 24 and 48h post-infection. (F) Top 12 canonical pathways showing progressive changes from 24 to 48 h post-infection. CD8+ T cell infiltration into lungs of SARS-CoV-2-infected mice. H&E staining of lungs of SARS-CoV-2 infected mice at day 7 post-infection reveal inflammation (A and C) associated with CD8+ T cell infiltration (B and D) as determined by immunofluorescent staining. Lymph node-like structures were also detected containing CD8+ T cells (E and F). Panels A, C, and E 10X magnification; panels B, D, and F 20X magnification.
CD8+ T cell infiltration into lungs of SARS-CoV-2-infected mice. H&E staining of lungs of SARS-CoV-2 infected mice at day 7 post-infection reveal inflammation (A and C) associated with CD8+ T cell infiltration (B and D) as determined by immunofluorescent staining. Lymph node-like structures were also detected containing CD8+ T cells (E and F). Panels A, C, and E 10X magnification; panels B, D, and F 20X magnification. Neurons are targets of infection within the brains of SARS-CoV-2 infected K18-hACE2 mice. Brains of SARS-CoV-2 infected mice at day 7 p.i. were analyzed to assess cellular targets of infection through in situ hybridization using RNAscope in situ hybridization using Spike-specific probes. (A) Cells within the cortex with neuron morphology were primary targets of infection; (B) high-power image of cells boxed in panel A show viral RNA present within cell body as well as extending down dendrites extending from the cell body. (C) Viral RNA was also detected in olfactory bulbs at day 7 p.i. (D) high-power image cells boxed in panel C reveal neurons in the mitral (open arrow) and glomerular (closed arrow) are infected by virus. Representative H&E images from the brains of infected K18-hACE2 mice at day 7 p.i. depicting (E) perivascular cuffing, (F) subventricular inflammation, and (G) leptomeningitis.
Neurons are targets of infection within the brains of SARS-CoV-2 infected K18-hACE2 mice. Brains of SARS-CoV-2 infected mice at day 7 p.i. were analyzed to assess cellular targets of infection through in situ hybridization using RNAscope in situ hybridization using Spike-specific probes. (A) Cells within the cortex with neuron morphology were primary targets of infection; (B) high-power image of cells boxed in panel A show viral RNA present within cell body as well as extending down dendrites extending from the cell body. (C) Viral RNA was also detected in olfactory bulbs at day 7 p.i. (D) high-power image cells boxed in panel C reveal neurons in the mitral (open arrow) and glomerular (closed arrow) are infected by virus. Representative H&E images from the brains of infected K18-hACE2 mice at day 7 p.i. depicting (E) perivascular cuffing, (F) subventricular inflammation, and (G) leptomeningitis.